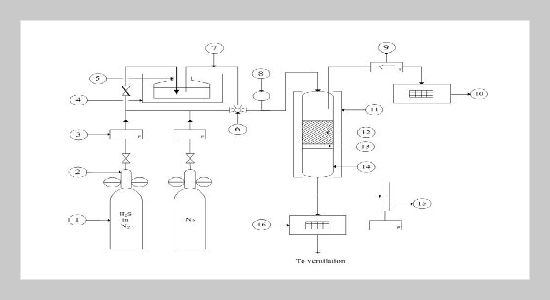
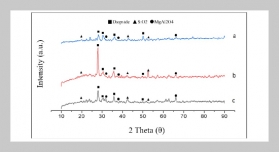
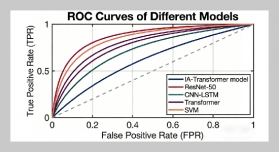
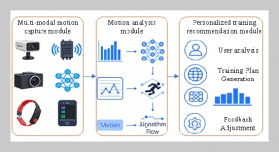
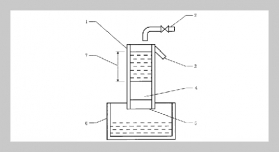
REFERENCES
- [1] Fenghua Shen, Jing Liu, Chenkai Gu, and Dawei Wu. Roles of Oxygen Functional Groups in Hydrogen Sulfide Adsorption on Activated Carbon Surface: A Density Functional Study. Industrial and Engineering Chemistry Research, 58(14):5526–5532, 2019.
- [2] Russamee Sitthikhankaew, Somrudee Predapitakkun, Ratanawan Kiattikomol, Sudhibhumi Pumhiran, Suttichai Assabumrungrat, and Navadol Laosiripojana. Comparative study of hydrogen sulfide adsorption by using alkaline impregnated activated carbons for hot fuel gas purification. In Energy Procedia, volume 9, pages 15–24, 2011.
- [3] Hui Sun Choo, Lee Chung Lau, Abdul Rahman Mohamed, and Keat Teong Lee. Hydrogen sulfide adsorption by alkaline impregnated coconut shell activated carbon. Journal of Engineering Science and Technology, 8(6):741–753, 2013.
- [4] Norhusna Mohamad Nor, Lee Chung Lau, Keat Teong Lee, and Abdul Rahman Mohamed. Synthesis of activated carbon from lignocellulosic biomass and its applications in air pollution control - A review, 2013.
- [5] Lee Chung Lau. Adsorption of hydrogen sulfide using palm shell activated carbon: an optimization study statistical analysis. International Journal of Research in Engineering and Technology, 02(08):302–311, 2013.
- [6] R Wojcieszak, M. Zieli ´ nski, S Monteverdi, and M M Bettahar. Study of nickel nanoparticles supported on activated carbon prepared by aqueous hydrazine reduction. Journal of Colloid and Interface Science, 299(1):238– 248, 2006.
- [7] Jiaojing Zhang, Lu Wang, Hualin Song, and Hualin Song. Adsorption of low-concentration H2S on manganese dioxide-loaded activated carbon. Research on Chemical Intermediates, 41(9):6087–6104, 2015.
- [8] Lee Chung Lau, Norhusna Mohamad Nor, Keat Teong Lee, and Abdul Rahman Mohamed. Selection of better synthesis route of CeO2/NaOH/PSAC for hydrogen sulphide removal from biogas. Journal of Environmental Chemical Engineering, 3(3):1522–1529, 2015.
- [9] N Mohamad Nor, L L Chung, Bassim H Hameed, S Sethupathi, and A R Mohamed. Effect of microwave heating variables on nitrogen-enriched palm shell activated carbon toward efficient hydrogen sulfide removal. In Solid State Phenomena, volume 280 SSP, pages 315–322, 2018.
- [10] Jia Guo, Ye Luo, Aik Chong Lua, Ru an Chi, Yan lin Chen, Xiu ting Bao, and Shou xin Xiang. Adsorption of hydrogen sulphide (H2S) by activated carbons derived from oil-palm shell. Carbon, 45(2):330–336, 2007.
- [11] Nurul Noramelya Zulkefli, Mohd Shahbudin Masdar, Wan Nor Roslam Wan Isahak, Jamaliah Md Jahim, Syahril Anuar Md Rejab, and Chew Chien Lye. Removal of hydrogen sulfide from a biogas mimic by using impregnated activated carbon adsorbent. PLoS ONE, 14(2), 2019.
- [12] Manisha Sharma, Raj K Vyas, and Kailash Singh. A review on reactive adsorption for potential environmental applications. Adsorption, 19(1):161–188, 2013.
- [13] Vincenzo Palma and Daniela Barba. Honeycomb V2O5-CeO2 catalysts for H2S abatement from biogas by direct selective oxidation to sulfur at low temperature. Chemical Engineering Transactions, 43:1957–1962,2015.
- [14] Valerio Paolini, Francesco Petracchini, Ettore Guerriero, Alessandro Bencini, and Serena Drigo. Biogas cleaning and upgrading with natural zeolites from stuffs. Environmental Technology (United Kingdom), 37(11):1418–1427, 2016.
- [15] A Daneshyar, M Ghaedi, M M Sabzehmeidani, and A Daneshyar. H2S adsorption onto Cu-Zn–Ni nanoparticles loaded activated carbon and Ni-Co nanoparticles loaded γ-Al2O3: Optimization and adsorption isotherms. Journal of Colloid and Interface Science, 490:553–561, 2017.
- [16] Abdul Hadi Abdullah, Ramli Mat, Azharin Shah Abd Aziz, and Fazlina Roslan. Use of kaolin as adsorbent for removal of hydrogen sulphide from biogas. Chemical Engineering Transactions, 56(2014):763–768, 2017.
- [17] Bharat Bajaj, Han Ik Joh, Seong Mu Jo, Ji Hye Park, Kwang Bok Yi, and Sungho Lee. Enhanced reactive H2S adsorption using carbon nanofibers supported with Cu/Cu x O nanoparticles. Applied Surface Science, 429:253–257, 2018.
- [18] JianWang, LongjiangWang, Huiling Fan, HuiWang, Yongfeng Hu, and Zhongde Wang. Highly porous copper oxide sorbent for H2S capture at ambient temperature. Fuel, 209(July):329–338, 2017.
- [19] Melody Kimi, Mohd Muazmil Hadi Jaidie, and Suh Cem Pang. Bimetallic Cu-Ni nanoparticles supported on activated carbon for catalytic oxidation of benzyl alcohol. Journal of Physics and Chemistry of Solids, 112:50–53, 2018.
- [20] Amvrosios G Georgiadis, Nikolaos D Charisiou, and Maria A Goula. Removal of hydrogen sulfide from various industrial gases: A review of the most promising adsorbing materials, 2020.
- [21] Jeerapan Tientong, Stephanie Garcia, Casey R Thurber, and Teresa D Golden. Synthesis of nickel and nickel hydroxide nanopowders by simplified chemical reduction. Journal of Nanotechnology, 2014, 2014.
- [22] Mykola Seredych, Denisa Hulicova-Jurcakova, Gao Qing Lu, and Teresa J Bandosz. Surface functional groups of carbons and the effects of their chemical character, density and accessibility to ions on electrochemical performance. Carbon, 46(11):1475–1488, 2008.
- [23] S Sumathi, S Bhatia, K T Lee, and A R Mohamed. Selection of best impregnated palm shell activated carbon (PSAC) for simultaneous removal of SO2 and NOx. Journal of Hazardous Materials, 176(1-3):1093–1096, 2010.
- [24] InduniWSiriwardane, Ranodhi Udangawa, Rohini M. de Silva, A R Kumarasinghe, Robert G Acres, Ananda Hettiarachchi, Gehan A.J. Amaratunga, and K. M.Nalin de Silva. Synthesis and characterization of nano magnesium oxide impregnated granular activated carbon composite for H2S removal applications. Materials and Design, 136:127–136, 2017.