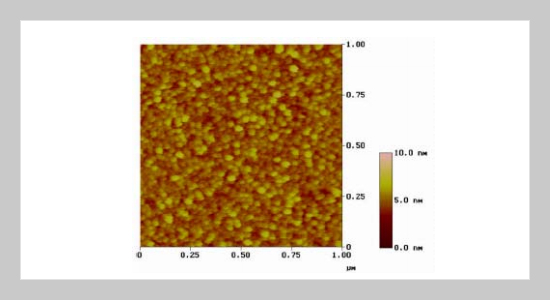
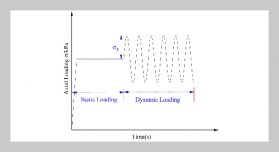
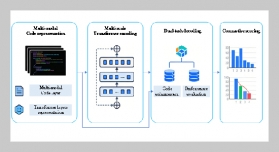
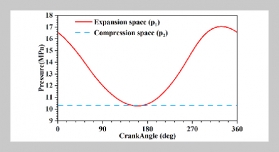
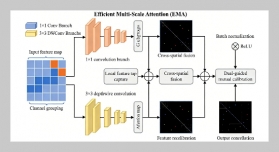
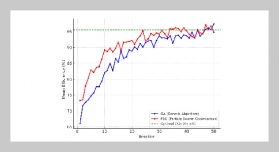
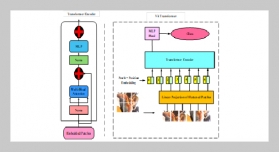
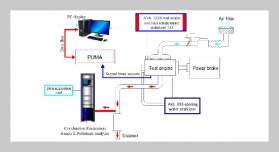
REFERENCES
- [1] Huang, Y., Duan, X., Q Wei and Leiber C. M., “Directed Assembly of One-Dimensional Nanostructures into Functional Networks,” Science, Vol. 291, pp. 630-33 (2001).
- [2] Gudiksen, M. S., Lauhon, L. J., Wang, J., Smith, D. C. and Leiber, C. M., “Growth of Nanowire Superlattice Structures for Nanoscale Photonics and Electronics,” Nature, Vol. 415, pp. 617 (2002).
- [3] Cui, Y. and Leiber, C. M., “Functional Nanoscale Electronic Devices Assembled Using Silicon Nanowire Building Blocks,” Science, Vol. 291, pp. 851-53 (2001).
- [4] Iijima, S., “Helical Microtubules of Graphitic Carbon,” Nature, Vol. 354, pp. 56 (1991).
- [5] Cui, Y., Lauhon, L. J., Gudiksen, M. S., Wang, J. and Lieber, C. M., “Diameter -Controlled Synthesis of Single-Crystal Silicon Nanowires,” Appl. Phys. Lett., Vol. 78, pp. 2214-16 (2001).
- [6] Burghard, Gu, G., M., Kim, G. T., Dusberg, G. S., Chiu, P. W., Krstic, V., Roth, S. and Han, W. Q., “Growth and Electrical Transport of Germanium Nanowires,” J. Appl. Phys., Vol. 90, pp. 5747-51(2001).
- [7] Duan, X., Huang, Y., Cui, Y., Wang, J., and Lieber, C. M., “Indium Phosphide Nanowires as Building Blocks for Nanoscale Electronic and Optoelectronic Devices,” Nature, Vol. 409, pp. 66 (2001).
- [8] Bai, Z. G., Yu, D. P., Zhang, H. Z., Ding, Y., Gai, S. Q., Hang, X. Z., Hiong, Q. L. and Feng, G. C., “Nano-Scale GeO2 Wires Synthesized by Physical Evaporation,” Chem. Phys. Lett., Vol. 303, pp. 311-14 (1999).
- [9] Huang, M. H., Wu, Y., Feick, H., Tran, N., Weber, E. and Yang, P., “Catalytic Growth of Zinc Oxide Nanowires by Vapor Transport,” Adv. Mater., Vol. 13, pp. 113-16 (2001).
- [10] Huang, M. H., Mao, S., Feick, H., Yan, H., Wu, Y., Kind, H., Weber, E., Russo, R. and Yang, P., “Room-Temperature Ultraviolet Nanowire Nanolasers,” Science, Vol. 292, pp. 1897-99 (2001).
- [11] Shi, G., Mo, C. M., Cai, W. L. and Zhang, L. D., “Photoluminescence of ZnO Nanoparticles in Alumina Membrane with Ordered Pore Arrays,” Solid State Commun. Vol. 115, pp. 253-56 (2000).
- [12] Wagner, R. S. and Ellis, W. C., “Vapor-Liquid-Solid Mechanism of Single Crystal Growth,” Appl. Phys. Lett., Vol. 4, pp. 89 (1964).
- [13] Wong, E. M. and Searson, P. C., “ZnO Quantum Particle Thin Films Fabricated by Electrophoretic Deposition,” Appl. Phys. Lett., Vol. 74, pp. 2939-41 (1999).
- [14] Tang, Z. K., Wong, G. K. L., Yu, P., Kawasaki, M., Ohtomo, A., Koinuma, H. and Segawa, Y., “Room-Temperature Ultraviolet Laser Emission from Self-Assembled ZnO Microcrystallite Thin Films,” Appl. Phys. Lett., Vol. 72, pp. 3270-72 (1998).
- [15] Li, J. F., Yao, L. Z., Mo, C. M., Cai, W. L., Zhang, Y. and Zhang, L. D., “Photoluminescence Enhancement of ZnO Nanocrystallites with BN Capsules,” J. Cryst. Growth., Vol. 223, pp. 535-38 (2001).
- [16] Vanheuseden, K., Warren, W. L., Seager, C. H., Tallant, D. R., Voigt, J. A. and Gnade, B. E., “Mechanisms Behind Green Photoluminescence in ZnO Phosphor Powders,” J. Appl. Phys., Vol. 79, pp. 7983-90 (1996).
- [17] Baker, H., Okamoto, H., Henry, S. D., Davidson, G. M., Fleming, M. A., Kacprzak, L. and Lampman, H. F., “ASM Handbook,” Vol. 3, pp. 276 (1987).
- [18] Wang, Y. P., Lee, W. I. and Tseng, T. Y., “Degradation Phenomena of Multilayer ZnO–Glass Varistors Studied by Deep Level Transient Spectroscopy,” Appl. Phys. Lett., Vol. 69, pp. 1807-09 (1996).
- [19] Lee, Y. S. and Tseng, T. Y., “Correlation of Grain Boundary Characteristics with Electrical Properties In ZnO - Glass Varistors,” J. Mater. Sci. Mater. In Electronics, Vol. 9, pp. 65-76 (1998).